Protein Translocation
Overview of Protein Translocation
Protein translocation is a sophisticated and critical biological process involving the movement of proteins from their synthesis site to their specific destinations. It encompasses mechanisms like co-translational and post-translational translocation, and involves specialized molecular machines and signals.
2. Mechanisms of Protein Translocation
a. Co-translational Translocation
Signal Sequence and SRP Recognition:
- Signal Sequence: The signal sequence is an N-terminal peptide, often rich in hydrophobic residues, that directs the nascent polypeptide to the ER.
- Signal Recognition Particle (SRP): SRP is a ribonucleoprotein complex consisting of an RNA component (SRP RNA) and six proteins (SRP54, SRP19, SRP68, SRP72, SRP9, SRP14). SRP54 binds to the signal sequence and the ribosome, pausing translation.
SRP-Receptor Interaction:
- Docking to ER Membrane: SRP binds to the SRP receptor (SR) on the ER membrane, which is composed of SRα and SRβ subunits. This interaction facilitates the transfer of the ribosome to the Sec61 translocon.
Sec61 Translocon:
- Structure: The Sec61 complex forms a protein-conducting channel in the ER membrane. It is composed of three main subunits (Sec61α, Sec61β, and Sec61γ) and associates with various auxiliary proteins such as TRAM (Translocon Associated Protein).
- Function: The Sec61 channel opens to allow the polypeptide chain to enter the ER lumen. It also undergoes conformational changes to accommodate different stages of protein transport.
Signal Peptide Cleavage and Protein Folding:
- Signal Peptidase: The signal peptide is cleaved off by signal peptidase in the ER lumen. The mature protein then folds into its functional conformation with the aid of ER chaperones like BiP (Binding Immunoglobulin Protein), calnexin, and calreticulin.
Quality Control:
- ERAD (ER-Associated Degradation): Misfolded proteins are targeted for degradation by the ubiquitin-proteasome system. They are retro-translocated to the cytosol and tagged with ubiquitin for proteasomal degradation.
b. Post-Translational Translocation
Mitochondrial Import:
- Precursor Proteins: Mitochondrial proteins are synthesized with mitochondrial targeting sequences (MTS) that are recognized by the TOM (Translocase of the Outer Membrane) complex.
- TOM Complex: TOM consists of multiple subunits, including TOM20 and TOM70 (receptors) and TOM40 (channel). It facilitates the initial import of precursor proteins into the intermembrane space.
- TIM Complexes: TIM23 and TIM22 complexes mediate the translocation of proteins across the inner mitochondrial membrane. TIM23, in conjunction with the membrane potential, translocates proteins into the matrix, while TIM22 handles the insertion of proteins into the inner membrane.
Nuclear Import:
- Nuclear Localization Signal (NLS): Proteins destined for the nucleus contain NLS, which is recognized by importins. Importins are also known as karyopherins.
- Nuclear Pore Complex (NPC): The NPC is a large structure composed of approximately 30 different nucleoporins (Nups). It forms a selective barrier that regulates the passage of proteins and RNAs between the nucleus and cytoplasm.
- Importins and Ran Cycle: Importins bind to the NLS-containing cargo in the cytoplasm, transport it through the NPC, and release it in the nucleus. The GTPase Ran binds to importin in the nucleus, facilitating the release of the cargo and recycling of importins back to the cytoplasm.
Chloroplast Import:
- TOC Complex: The Translocon at the Outer Chloroplast membrane (TOC) complex consists of TOC75 (a channel-forming component) and TOC159/TOC33 (receptors).
- TIC Complex: The Translocon at the Inner Chloroplast membrane (TIC) complex helps in the translocation of proteins across the inner membrane into the stroma. The TIC complex includes TIC110 and TIC40.
c. Vesicular Transport
Vesicle Formation:
- Coat Proteins: Vesicle formation involves coat proteins like clathrin (for transport between the Golgi and plasma membrane) and COPI/COPII (for ER-Golgi transport). Clathrin forms a triskelion shape and coats the vesicle, while COPI and COPII form coats on the cytoplasmic surface of the vesicle.
Vesicle Transport:
- Motor Proteins: Vesicles are transported along cytoskeletal tracks (microtubules) by motor proteins such as kinesins and dyneins. These motor proteins use ATP to move vesicles to their target membranes.
- Anterograde transport refers to the movement of cargo from the endoplasmic reticulum (ER) towards the cell membrane or other destinations like the Golgi apparatus and lysosomes. This transport is essential for delivering newly synthesized proteins and lipids to their functional locations.
- Retrograde transport involves the movement of cargo from the plasma membrane or Golgi back to the ER or other earlier compartments. This process is crucial for recycling components, retrieving misfolded proteins, and regulating cellular homeostasis.
Vesicle Fusion:
- SNARE Proteins: The fusion of vesicles with target membranes is mediated by SNARE proteins. v-SNAREs (vesicle-SNAREs) on the vesicle and t-SNAREs (target-SNAREs) on the target membrane interact to facilitate membrane fusion and cargo release.
3. Advanced Aspects of Protein Translocation
a. Signal Recognition and Integration:
- Signal Sequences and Sorting Signals: Proteins contain various signal sequences and sorting signals that determine their final destination. The specificity of signal recognition by cellular machinery ensures correct protein localization.
b. Post-Translational Modifications:
- Phosphorylation, Glycosylation, and Acetylation: These modifications can affect protein stability, localization, and function. For instance, phosphorylation of a nuclear localization signal can regulate nuclear import.
c. Diseases Related to Protein Translocation:
- Alpha-1 Antitrypsin Deficiency: Caused by defective folding and translocation of the alpha-1 antitrypsin protein, leading to liver disease and lung damage.
- Hereditary Spastic Paraplegia: Associated with mutations affecting protein import into mitochondria, resulting in neurodegenerative symptoms.
d. Experimental Techniques for Studying Protein Translocation:
- Live-Cell Imaging: Techniques such as fluorescence recovery after photobleaching (FRAP) and fluorescence resonance energy transfer (FRET) help study dynamic aspects of protein localization and movement in real-time.
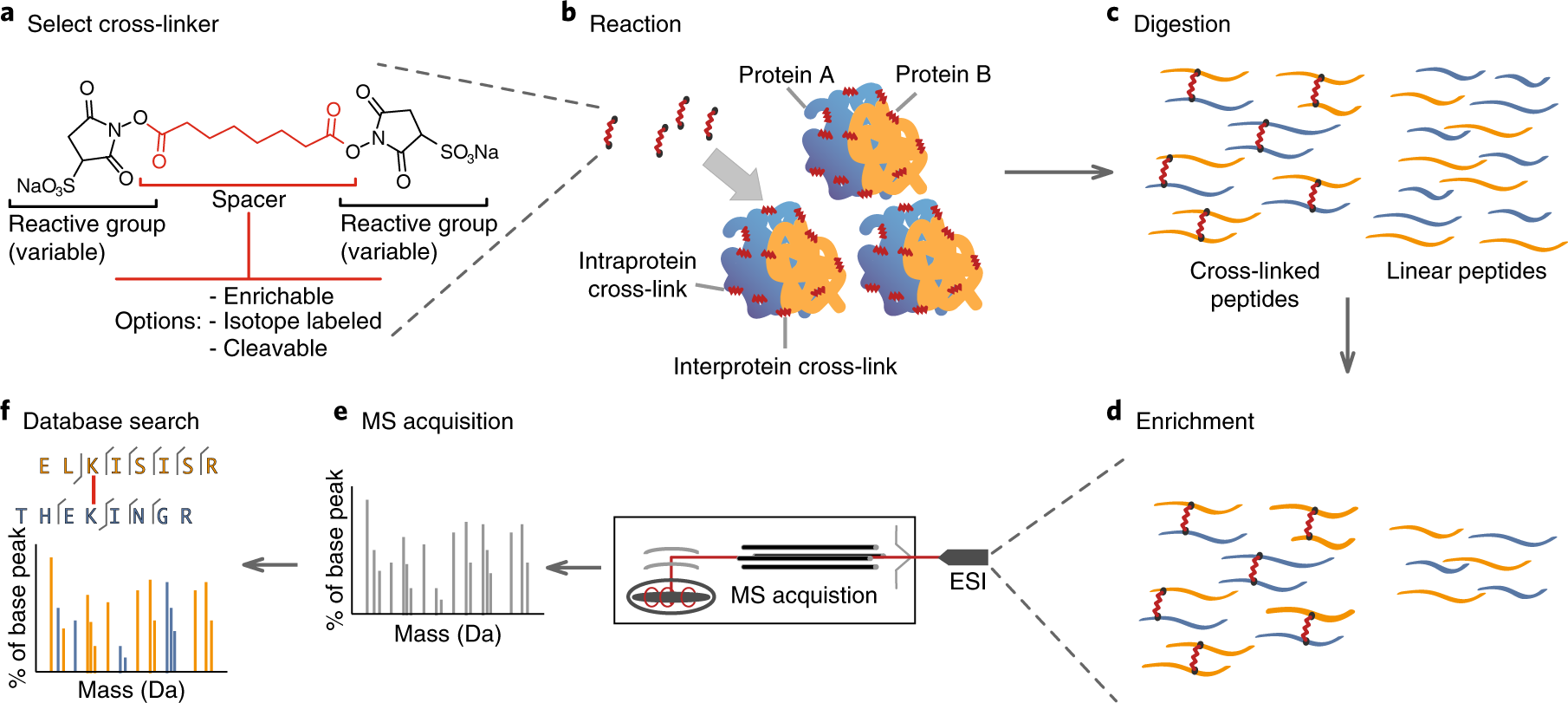
- Cross-Linking and Mass Spectrometry: These methods are used to study protein-protein interactions and the identification of transient protein complexes involved in translocation.
Please, give your suggetions in comment section.
Follow our blogs for more updates and protocols: https://iammolecularbiologist.blogspot.com/









0 Comments:
Post a Comment