1. Introduction to Proteins
- Definition: Proteins are macromolecules consisting of one or more long chains of amino acid residues.
- Structure: Proteins are composed of carbon (C), hydrogen (H), oxygen (O), nitrogen (N), and occasionally sulfur (S) atoms. They are synthesized based on the genetic code.
2. Amino Acids
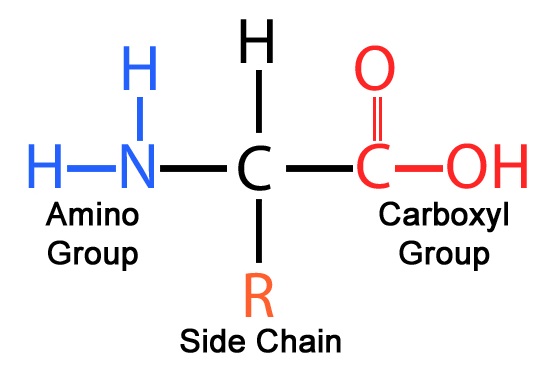
- Basic Structure: Each amino acid contains:
- A central alpha carbon (Cα)
- An amino group (−NH2)
- A carboxyl group (−COOH)
- A hydrogen atom (H)
- A distinctive side chain (R group), which determines the properties and functions of the amino acid.
- Classification: Amino acids are categorized based on the properties of their side chains:
- Nonpolar, aliphatic: Glycine, alanine, valine, leucine, isoleucine, methionine.
- Aromatic: Phenylalanine, tyrosine, tryptophan.
- Polar, uncharged: Serine, threonine, cysteine, proline, asparagine, glutamine.
- Positively charged (basic): Lysine, arginine, histidine.
- Negatively charged (acidic): Aspartate, glutamate.
3. Protein Structure Levels
- Primary Structure: The linear sequence of amino acids in a polypeptide chain, determined by mRNA.
- Secondary Structure: Local folding patterns within the polypeptide stabilized by hydrogen bonds.
- Alpha Helix: Right-handed coil stabilized by hydrogen bonds between the backbone carbonyl oxygen and amide hydrogen atoms four residues apart.
- Beta Sheet: Strands connected laterally by at least two or three backbone hydrogen bonds, forming a sheet-like array.
- Tertiary Structure: The overall three-dimensional shape of a polypeptide, stabilized by interactions between R groups, including hydrogen bonds, ionic bonds, hydrophobic interactions, and disulfide bridges (covalent bonds between cysteine residues).
- Quaternary Structure: The structure formed by multiple polypeptide chains (subunits), which may be identical (homomers) or different (heteromers). Hemoglobin is an example of a protein with quaternary structure.
4. Protein Functions
- Enzymatic Activity: Catalysts that accelerate biochemical reactions by lowering activation energy. Examples include:
- Hydrolases: Break bonds by hydrolysis (e.g., proteases).
- Ligases: Join two molecules together (e.g., DNA ligase).
- Oxidoreductases: Catalyze oxidation-reduction reactions (e.g., oxidases).
- Structural Role: Provide cellular and tissue structure, as in:
- Collagen: Found in connective tissues.
- Keratin: Key component of hair, nails, and the epidermis.
- Transport and Storage: Transport molecules or store ions and molecules.
- Hemoglobin: Transports oxygen in the blood.
- Ferritin: Stores iron in the liver.
- Signal Transduction: Function as receptors and hormones in cellular communication.
- Insulin: Regulates glucose metabolism.
- G-protein-coupled receptors (GPCRs): Transmit signals across cell membranes.
- Immune Response: Antibodies (immunoglobulins) identify and neutralize pathogens.
5. Protein Synthesis
- Transcription: Synthesis of mRNA from a DNA template in the nucleus. Key steps include:
- Initiation: RNA polymerase binds to the promoter region of the gene.
- Elongation: RNA polymerase synthesizes mRNA by adding nucleotides complementary to the DNA template.
- Termination: Transcription ends when RNA polymerase reaches a terminator sequence.
- Translation: The mRNA is translated into a polypeptide chain at the ribosome.
- Initiation: The ribosome assembles around the mRNA and the first tRNA.
- Elongation: tRNAs bring amino acids to the ribosome, where they are added to the growing polypeptide chain.
- Termination: The process ends when a stop codon is reached.
6. Protein Digestion and Absorption
- Digestion: Begins in the stomach with pepsin and continues in the small intestine with proteases (e.g., trypsin, chymotrypsin, carboxypeptidase).
- Absorption: Amino acids and small peptides are absorbed by enterocytes in the small intestine via active transport and facilitated diffusion.
7. Protein Metabolism
- Anabolism: Biosynthesis of proteins from amino acids.
- Transamination: Transfer of an amino group from one amino acid to a keto acid, forming a new amino acid.
- Aminoacyl-tRNA Synthetase: Enzyme that attaches amino acids to their corresponding tRNA.
- Catabolism: Breakdown of proteins into amino acids and further into ammonia, urea, and keto acids.
- Deamination: Removal of an amino group from an amino acid.
- Urea Cycle: Converts toxic ammonia to urea for excretion.
8. Protein Requirements and Sources
- Daily Requirements: Varies by individual factors. The RDA is approximately 0.8 grams per kilogram of body weight for adults.
- Sources:
- High-Quality Proteins (Complete Proteins): Provide all essential amino acids in sufficient quantities (e.g., meat, eggs, dairy).
- Lower-Quality Proteins (Incomplete Proteins): Lack one or more essential amino acids (e.g., most plant-based sources like grains, nuts, and legumes).
9. Protein Deficiency
- Kwashiorkor: Severe protein deficiency characterized by edema, an enlarged liver, and dermatosis.
- Marasmus: Severe calorie and protein deficiency, leading to significant wasting and stunted growth.
10. Protein Quality
- Biological Value (BV): Measures the proportion of absorbed protein incorporated into body proteins. High BV proteins provide a greater proportion of utilized amino acids.
- Protein Digestibility Corrected Amino Acid Score (PDCAAS): Evaluates protein quality based on amino acid requirements and digestibility.
- Net Protein Utilization (NPU): Ratio of amino acids converted to proteins to the amino acids supplied.
11. Protein and Health
- Muscle Maintenance and Growth: Essential for muscle protein synthesis, repair, and maintenance. Influenced by factors like exercise and protein intake.
- Immune Function: Essential for the synthesis of immunoglobulins and other immune system components.
- Hormone Production: Many hormones, such as insulin and growth hormone, are protein-based.
12. Protein Supplements
- Types:
- Whey Protein: Fast-digesting, high in essential amino acids, especially leucine.
- Casein Protein: Slow-digesting, provides a steady release of amino acids.
- Plant-Based Proteins: Derived from soy, pea, rice, etc., suitable for vegetarians and vegans.
- Uses: Often used to supplement dietary protein intake for muscle building, recovery, and other health benefits.

:max_bytes(150000):strip_icc():format(webp)/protein-structure-373563_final11-5c81967f46e0fb00012c667d.png)





0 Comments:
Post a Comment